By Donnie Yance
We read with interest the study by Ayoade et al.1 examining the association between complementary and alternative medicine (CAM) use and survival in breast cancer patients. While the authors present important data from a large national cohort, we believe the study’s fundamental approach to categorizing CAM presents significant methodological limitations that substantially compromise the clinical translatability of these findings.
The Problem of Definitional Heterogeneity
The central limitation lies in how CAM is operationalized in the National Cancer Database (NCDB) and consequently in this analysis: as “therapy administered by nonmedical personnel.” This definition creates an extraordinarily heterogeneous category that conflates interventions with vastly different mechanisms, evidence bases, and clinical applications. Under this umbrella fall experimental unconventional drugs, unproven treatments, evidence-based herbal medicines, therapeutic nutrition interventions, mind-body practices, and numerous other modalities. This “everything that isn’t conventional medicine” approach obscures far more than it reveals.
Consider the clinical and biological differences between a patient who refuses chemotherapy in favor of high-dose intravenous vitamin C administered at a non-accredited facility versus a patient who completes all recommended conventional treatments while also working with a registered dietitian on an anti-inflammatory Mediterranean diet and taking curcumin supplements with established pharmacokinetic profiles. Both would be categorized identically as “CAM users” in this dataset, yet these scenarios represent fundamentally different clinical decisions with presumably different outcomes.
The problem is further compounded by the inclusion of “experimental unconventional drugs” within the CAM category. Experimental agents—whether conventional or unconventional—represent a distinct category of treatment that should be analyzed separately from established complementary approaches. Lumping experimental drugs together with evidence-based nutritional interventions and herbal medicines creates a methodologically untenable comparison group.
Evidence for Nutrition and Herbal Medicine: A Different Story
When examined with appropriate granularity, the evidence base for certain nutritional interventions and herbal medicines in cancer care is substantial and growing. Multiple systematic reviews and meta-analyses have demonstrated that evidence-based nutritional support improves treatment tolerance, reduces treatment-related toxicity, and may improve survival outcomes in cancer patients. For instance, L-glutamine,2,3 tocotrinols,4,5 specific fatty acids such as Gama linolenic acid (GLA),6 and EPA/DHA rich omega-3 fatty acid7,8,9,10 and zinc11,12 supplementation has been shown to reduce chemotherapy-induced peripheral neuropathy, improve quality of life and increase lifespan in cancer patients. Anabolic wholefood nutrition with high-quality Whey Protein Concentrate13,14 and Chlorella is associated with better treatment completion rates and outcomes.
Similarly, certain herbal medicines have robust evidence supporting their use as complementary therapies. Turkey Tail mushroom for general immune support, Ginger for chemotherapy-induced nausea, American ginseng for cancer-related fatigue, and various adaptogens for treatment-related side effects have demonstrated efficacy in randomized controlled trials. These interventions, when used alongside—not instead of—conventional therapy, represent evidence-based supportive care rather than “alternative” medicine.
The overwhelming evidence supporting proper nutrition during cancer treatment makes it particularly problematic to categorize nutritional interventions within a broad CAM category that shows negative outcomes. This conflation risks discouraging patients from pursuing evidence-based nutritional support that could improve their treatment tolerance and outcomes.
Treatment Avoidance Versus Complementary Harm
A critical finding in the study actually supports the argument that the observed harm is not from complementary modalities themselves, but rather from incomplete conventional treatment. The authors report that patients in the combination cohort (CAM plus conventional therapy) were significantly less likely to receive endocrine therapy (40.7% vs 65.2% in stage II disease) and radiation therapy (59.5% vs 36.6% in stage II disease) compared to patients receiving conventional therapy alone.
This finding suggests that the increased mortality in the combination group is not attributable to harm caused by the complementary modalities themselves, but rather to the refusal or delay of conventional treatments. The CAM use appears to be a marker for treatment avoidance rather than a causative factor in mortality. Patients who fully complete conventional therapy while also using evidence-based complementary approaches have more positive outcomes than those who use CAM as a justification for refusing or delaying conventional treatment, yet the current analysis cannot distinguish between these opposite scenarios.
This distinction has profound clinical implications. The appropriate clinical message is not “avoid all complementary approaches,” but rather “complete all recommended conventional treatments.” A patient who undergoes surgery, completes chemotherapy and radiation, adheres to endocrine therapy, and also takes curcumin supplements and follows an anti-inflammatory diet is making fundamentally different choices than a patient who refuses radiation in favor of coffee enemas, yet both are categorized as CAM users in this analysis.
The Need for Nuanced Research
To generate clinically actionable evidence, future research must disaggregate the heterogeneous CAM category into meaningful subcategories. At minimum, studies should distinguish between:
- Evidence-based nutritional interventions (therapeutic diets, specific supplements with established mechanisms)
- Traditional herbal medicines with documented pharmacology
- Mind-body practices (meditation, yoga, acupuncture)
- Unproven or experimental treatments
- Practices used instead of versus alongside of conventional therapy
Additionally, analyses should control for completion of conventional therapy as a mediating variable, allowing researchers to determine whether complementary approaches used in addition to—rather than instead of—full conventional treatment are associated with different outcomes.
Benefits of Evidence-Based Unitive Medicine
While Ayoade et al. provide valuable epidemiologic data on treatment patterns, the study’s reliance on an overly broad and heterogeneous definition of CAM significantly limits clinical translatability and obscures critical distinctions. The evidence base (documentation) for evidence-based nutritional and herbal interventions as complementary to conventional therapy is substantial and should not ever be conflated with unproven expensive treatments—such as unregulated intravenous therapies, chelation devices, and hyperbaric oxygen protocols—that patients pursue at specialized clinics at considerable financial and temporal cost, yet without demonstrated survival benefit. More nuanced research that distinguishes between evidence-based complementary modalities and unproven experimental treatments, while controlling for completion of conventional therapy, is essential. Equally important is enhanced patient education that clarifies these distinctions, enabling informed decision-making and ensuring that patients complete proven conventional treatments while safely incorporating evidence-based supportive care.
This distinction is not merely theoretical. A published retrospective case study15 of 33 breast cancer patients at Mederi Center who received evidence-based integrative nutritional, botanical, and lifestyle interventions alongside conventional treatment demonstrated meaningful improvements across multiple quality of life domains, including sleep quality, anxiety reduction, empowerment, and overall wellbeing—providing concrete evidence that well-designed complementary approaches, when used to enhance rather than replace conventional therapy, can produce measurable positive patient outcomes. Such evidence underscores the critical need to move beyond categorical dismissal of all CAM modalities and instead develop research frameworks that distinguish evidence-based integrative care from unproven experimental treatments.
The Mederi Care Difference
Mederi Care embraces the complexity of diseases by supporting the general philosophy of building robustness, auto-regulation and addressing underlying dysregulating biological networks.
A mechanistic understanding of cancer requires a detailed mapping of the underlying perturbations across biological networks.16
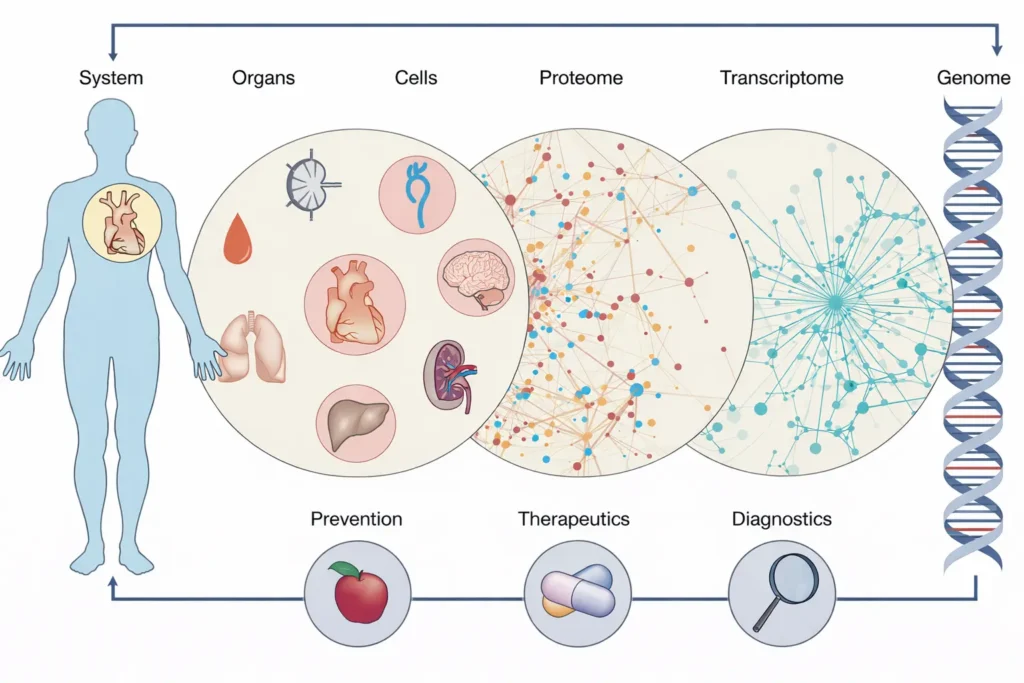
The Mederi-Care approach is to improve patient outcomes by supporting a patient’s autoregulatory capacity at the organ system, cellular, and molecular levels. This is accomplished through the Mederi Care toolboxes, specifically botanical and nutritional medicine, which is applied in a gentle, synergistic way. Botanical and nutritional medicine practiced within Mederi Care is primarily directed at enhancement of ‘Self-regulating Internal Community Networks,’ supporting and even directing, while allowing the freedom to improvise.
Herbal medicines are often combinations of botanical extracts that have additive or synergistic effects. For example, combining four herbal extracts (S. baicalensis, D. morifolium, G. uralensis and R. rubescens) significantly enhanced their activity compared with extracts alone in a prostate cancer model.17
It is important not to confuse synergistic effects with additive effects. Synergy occurs when two or more drugs/compounds are combined to produce a total effect that is greater than the sum of the individual agents while an additive effect is an add up of individual effects where each individual agent is not affecting the others (no interactions).18
The synergy of biological effects of plants in medicine is well documented, and encompasses synergistic multitarget effects, physicochemical effects based on improved solubility, antagonization of resistance mechanisms, and elimination or neutralization of toxic substances.19 As such, multi-combination and/or multi-system low dose medications, preferably of natural origin, are well suited for the bioregulatory medical approach and offer the potential for a graded response to treatment.20
Generally speaking, herbal and nutritional medicine within this model exhibits four fundamental advantages of a multicomponent, combinatorial strategy over a single-component strategy:
1. Synergistic effects target a wider range of information flow in disease-related biological networks;
2. Modest modulation allows for more efficient control of biological networks;
3. Lower concentrations ensure higher safety of the whole combination;
4. Drug resistance is much less probable.21
Mederi Care exemplifies the kind of granular, evidence-based approach to herbal and nutritional medicine that this rebuttal calls for—moving beyond undifferentiated “CAM” categorization to specific, mechanistically informed interventions used alongside conventional cancer treatment. Rather than representing treatment avoidance or rejection of oncologic care, Mederi Care emphasizes the practice of ‘health-promoting medicine’ as the foundation for long-term healing, with the primary goal of bringing the body into harmonious balance through nontoxic or low-toxicity, target-specific, disease-suppressing agents.
This model directly embodies the four multicomponent advantages outlined above: (1) synergistic herbal and nutritional formulations that target multiple nodes in disease-related biological networks; (2) modest modulation through naturally phytochemically complex medicines that allow efficient network control; (3) low-dose combinations that ensure higher safety profiles; and (4) multi-target approaches that reduce the probability of resistance mechanisms. These formulations are applied in conjunction with dietary therapeutics and other specific therapies as clinically indicated, providing the definitional clarity and clinical actionability absent from broad “CAM user” classifications.
Mederi Care represents truly Unitive Medicine—integrating evidence-based botanical and nutritional interventions with conventional oncologic care rather than substituting for it.
For more information about Mederi Care, including training programs for health care professionals, please go to Mederifoundation.org and Donnieyance.com.
- Ayoade OF, Caturegli G, Canavan ME, Resio BJ, Berger ER, Boffa DJ. Use of Complementary and Alternative Medicine in the Management of Breast Cancer. JAMA Netw Open. 2026 Mar 2;9(3):e260337. doi: 10.1001/jamanetworkopen.2026.0337. PMID: 41770560; PMCID: PMC12954545.
- Chen L, Wang D, Meng C, Sun H, Li R, Miao G, Liu P. Glutamine prevents diarrhea in colorectal cancer patients undergoing chemotherapy or chemoradiotherapy: a meta-analysis. BMC Gastroenterol. 2025 Oct 6;25(1):697. doi: 10.1186/s12876-025-04308-w. PMID: 41053591; PMCID: PMC12502349.
- Vahdat L, et al. Reduction of paclitael-induced peripheral neuropathy with glutamine. Clin Cancer Res 2001;1;7:1192- 1197.
- Sailo BL, Chauhan S, Hegde M, Girisa S, Alqahtani MS, Abbas M, Goel A, Sethi G, Kunnumakkara AB. Therapeutic potential of tocotrienols as chemosensitizers in cancer therapy. Phytother Res. 2025 Apr;39(4):1694-1720. doi: 10.1002/ptr.8131. Epub 2024 Feb 14. PMID: 38353331.
- Eitsuka T, Tatewaki N, Nishida H, Nakagawa K, Miyazawa T. Synergistic Anticancer Effect of Tocotrienol Combined with Chemotherapeutic Agents or Dietary Components: A Review. Int J Mol Sci. 2016 Sep 22;17(10):1605. doi: 10.3390/ijms17101605. PMID: 27669218; PMCID: PMC5085638.
- Arsic A, Krstic P, Paunovic M, Nedovic J, Jakovljevic V, Vucic V. Anti-inflammatory effect of combining fish oil and evening primrose oil supplementation on breast cancer patients undergoing chemotherapy: a randomized placebo-controlled trial. Sci Rep. 2023 Apr 20;13(1):6449. doi: 10.1038/s41598-023-28411-8. PMID: 37081029; PMCID: PMC10119093.
- Murphy RA, Mourtzakis M, Chu QSC, et al. Supplementation with fish oil increases first-line chemotherapy efficacy in patients with advanced nonsmall cell lung cancer. Cancer 2011;117:3774-80.
- Bougnoux P, et al. Improving outcome of chemotherapy of metastatic breast cancer by docosahexaenoic acid: a phase II trial. Br J Cancer. 2009 December 15; 101(12): 1978–1985.
- Darwito D, Dharmana E, Riwanto I, Budijitno S, Suwardjo S, Purnomo J, Widodo I, Ghozali A, Aryandono T, Anwar SL. Effects of Omega-3 Supplementation on Ki-67 and VEGF Expression Levels and Clinical Outcomes of Locally Advanced Breast Cancer Patients Treated with Neoadjuvant CAF Chemotherapy: A Randomized Controlled Trial Report, Asian Pac J Cancer Prev. 2019 Mar 26;20(3):911-916.
- Biondo PD, Brindley DN, Sawyer MB, Field CJ. The potential for treatment with dietary long-chain polyunsaturated n-3 fatty acids during chemotherapy. J Nutr Biochem. 2008 Dec;19(12):787-96. doi: 10.1016/j.jnutbio.2008.02.003. Epub 2008 Jul 7. PMID: 18602809.
- Kocdor H, Ates H, Aydin S, Cehreli R, Soyarat F, Kemanli P, Harmanci D, Cengiz H, Kocdor MA. Zinc supplementation induces apoptosis and enhances antitumor efficacy of docetaxel in non-small-cell lung cancer. Drug Des Devel Ther. 2015 Jul 27;9:3899-909. doi: 10.2147/DDDT.S87662. PMID: 26251569; PMCID: PMC4524380.
- Ribeiro SMF, Braga CBM, Peria FM, Martinez EZ, Rocha JJRD, Cunha SFC, Effects of zinc supplementation on fatigue and quality of life in patients with colorectal cancer, Einstein (Sao Paulo). 2017 Jan-Mar;15(1):24-28. doi: 10.1590/S1679-45082017AO3830.
- Cereda E, Turri A, Klersy C, Cappello S, Ferrari A, Filippi AR, Brugnatelli S, Caraccia M, Chiellino S, Borioli V, Monaco T, Stella GM, Arcaini L, Benazzo M, Grugnetti G, Pedrazzoli P, Caccialanza R. Whey protein isolate supplementation improves body composition, muscle strength, and treatment tolerance in malnourished advanced cancer patients undergoing chemotherapy. Cancer Med. 2019 Nov;8(16):6923-6932. doi: 10.1002/cam4.2517. Epub 2019 Sep 30. PMID: 31568698; PMCID: PMC6853834.
- Bumrungpert A, Pavadhgul P, Nunthanawanich P, Sirikanchanarod A, Adulbhan A. Whey Protein Supplementation Improves Nutritional Status, Glutathione Levels, and Immune Function in Cancer Patients: A Randomized, Double-Blind Controlled Trial. J Med Food. 2018 Jun;21(6):612-616. doi: 10.1089/jmf.2017.4080. Epub 2018 Mar 12. PMID: 29565716.
- Niemeyer KJ. Self-Assessment of Change and Experiences of Persons with Breast Cancer Using Unitive Whole-Person Integrative Health. Integrative and Complementary Therapies. 2022;28(5):221-231. doi:10.1089/ict.2022.29040.kjn
- Schmidt, H.H.H.W., Menche, J. The regulatory network architecture of cardiometabolic diseases. Nat Genet 54, 2–3 (2022). https://doi.org/10.1038/s41588-021-00994-w
- Adams LS, Seeram NP, Hardy ML, Carpenter C, Heber D. Analysis of the interactions of botanical extract combinations against the viability of prostate cancer cell lines. Evid Based Complement Alternat Med. 2006 Mar;3(1):117-24. doi: 10.1093/ecam/nel001.
- Zhou, Xian et al. “Synergistic Effects of Chinese Herbal Medicine: A Comprehensive Review of Methodology and Current Research.” Frontiers in pharmacology vol. 7 201. 12 Jul. 2016, doi:10.3389/fphar.2016.00201
- Wagner H. 2011. Synergy research: Approaching a new generation of phytopharmaceuticals. Fitoterapia. 82(1):34–37
- Bioregulatory Systems Medicine. White Paper. Published February 2019 – Copyright © Biologische Heilmittel Heel GmbH. Rights Reserved
- Kong DX, Li XJ, Zhang HY. 2009. Where is the hope for drug discovery? Let history tell the future