Natural Compounds for Beating Dormant Breast Cancer Stem Cells

By Donnie Yance October is Breast Cancer Awareness Month. This review examines one of breast cancer’s most challenging aspects: dormant disseminated tumor cells (DTCs) that fuel cancer recurrence. Despite initial treatment success, 25-45% of breast cancer patients relapse months or years later with fatal secondary or metastatic disease, with dormancy being the key culprit behind […]
Modified Citrus Pectin: One Of Nature’s Answers To Cancer Cells’ Spread

By Donnie Yance Cancer becomes most dangerous when it spreads from its original location to other parts of the body—a process called metastasis. This spreading relies on certain proteins called lectins, which act like molecular “velcro” helping cancer cells stick together and communicate with each other. 1 Using modified citrus pectin for cancer is one […]
Parmigiano Reggiano: The King of Cheeses

by Donnie Yance Parmigiano Reggiano, a cheese to which I am somewhat addicted, offers a delicious sharp, nutty, and slightly salty flavor that has earned it the nickname “King of Cheeses” among culinary enthusiasts worldwide. The Health Paradox of Cheese Cheese is a nutrient-dense fermented dairy product consumed worldwide, though its health effects remain controversial. […]
The Benefits of Baking Soda For Cancer?

By Donnie Yance Is baking soda for cancer a valid idea? Cancer (The Cancer Energy/Intelligence) alters the tumor microenvironment in several ways, and one of them is by creating an acidic zone around the tumor that promotes cancer progression and treatment resistance. Recent research suggests that sodium bicarbonate (baking soda) or potassium bicarbonate supplementation can […]
Prostate Cancer: The Pitfalls of Conventional Assessments

By Donnie Yance Questioning Conventional Screening Approaches According to the United States Preventive Services Task Force (USPSTF) analysis of two major randomized clinical trials, routine Prostate-specific antigen (PSA) screening prevents approximately one prostate cancer-related death per 1,000 men screened.¹ This sobering statistic challenges the historical perception of PSA screening as an essential preventive measure. Moreover, […]
When Sound Waves Become Healing Waves: Understanding Histotripsy

By Donnie Yance As a musician and composer, I’ve always been fascinated by the power of sound. We know sound waves can shatter glass, create beautiful music, and even help babies develop in the womb. But what if I told you that precisely controlled sound waves could also destroy cancer cells while leaving surrounding healthy […]
COVID-19 Infection Triggers An Anti-Tumor Immune Response In Some Cancer Patients. Part 2: Traditional Herbal Medicine- The Immune System’s Secret Weapon

By Donnie Yance Traditional Herbal Medicine (THM) is very different from using isolated plant compounds. Using isolates to treat cancer is a more allopathic approach and not at all part of a holistic whole-systems model based in THM practice. THM is based on a concept of whole plants having synergy as well as intelligence (Plant […]
What You Need To Know About IV Vitamin C and Cancer Seen Through The Lens Of Mederi-Care
By Donnie Yance “The noblest foundation for medicine is love. It is love that teaches us the art of healing. Without love true healing can never be born.” – Paracelsus Intravenous (IV) nutrient therapy, which most often includes vitamins, minerals, and sometimes botanical isolates has become very popular these days. IV vitamin C and cancer […]
Curcumin in Combination with Chemotherapy: A Positive Interaction
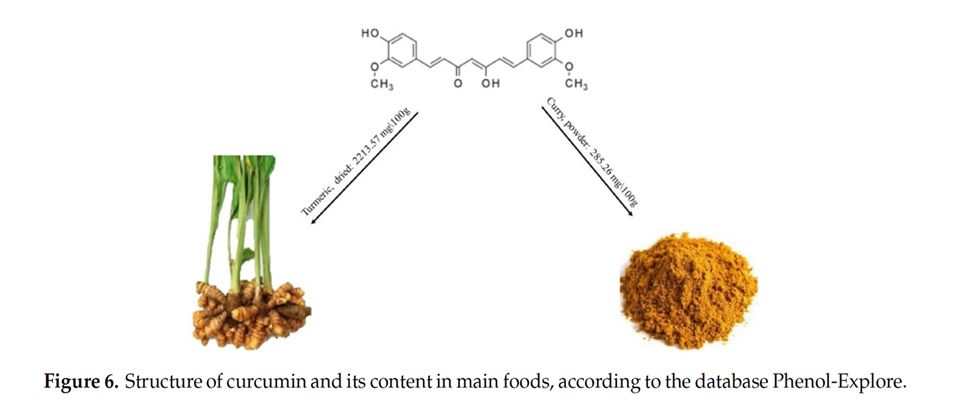
Natural compounds, including curcumin, resveratrol, EGCG, and β-glucan have shown synergistic promising immune-modulating, anti-tumor, and chemo-potentiating effects.
The results of these clinical studies are conclusive, and these studies have established a good foundation for further research focusing on implementing curcumin along with other botanical compounds in clinical oncology. It’s important to note, however, that I never use curcumin as a soloist! I always use a formula that combines curcumin with EGCG, resveratrol, grape seed extract, quercetin, and other botanical extracts. This provides a harmonious approach that best supports healing.
